| Necturus maculosus | |||||||||
| Mudpuppy | |||||||||

|
|
||||||||
| Necturus m. maculosus |
Description
Mudpuppies are large aquatic salamanders found in the eastern United States and Canada. Belonging to the family Proteidae, the genus Necturus currently consists of 5 species. Mudpuppies are often confused with axolotls (Ambystoma mexicanum), sirens (Siren spp.) or with larval tiger salamanders (primarily Ambystoma mavortium), which are also known as waterdogs. Mudpuppies are easily distinguished from other aquatic salamanders, as they possess 4 toes on the hind feet, whereas most other salamanders possess 5. The different species of Necturus differ greatly in size, with the largest (N. maculosus) reaching lengths up to 49 cm (19 in), and the smallest (N. punctatus) maturing at 11.5-19 cm (4.5-7.5 in) (Petranka, 1998).
Necturus maculosus are generally dark colored, ranging through a number of shades of brown to black. Typically, they exhibit dark spots on a lighter background, though this is not always the case. The venter is lighter in color, ranging from light brown to white or yellow. N. m. louisianensis have a light yellowish brown to tan dorsum (Petranka, 1998). Gills are bushy and red. All four limbs are fully functional and are not reduced like those of sirens or amphiumas. All four feet have four toes. The tail is laterally compressed and rounded and well adapted for swimming. However, they often use their legs for locomotion, walking slowly along the bottom. Necturus have a large, stout body with a wide, flattened head. The head has two small eyes, usually with a dark stripe extending across them. Juveniles are smaller, and exhibit light-colored stripes down the entire length of the body; in most species, these stripes are lost at maturity.
Unless otherwise stated, all further references to Necturus refer to N. maculosus. Care for other species is similar.
 N. m. maculosus |
 N. m. maculosus |
Natural Range and Habitat
N. maculosus are found in most states in the eastern half of the United States. Their range extends into the southern parts of the eastern Canadian provinces. There are two subspecies: N. m. maculosus in the north, and N. m. lousianensis in the southwestern portion of the range (most of Arkansas and parts of the surrounding states).
Necturus live in rivers, lakes, and other permanent water sources. One study suggests that they prefer water depth ranges of 21-35 cm (8-14 in) and 86-95 cm (34-37 in) (Sajdak, 1982). However, they are frequently found at other depths and have been found as deep as 27 m (88 ft). They prefer hard cover such as rocks, logs, and overhangs, but will also utilize debris, reeds, mud, stream banks, and other areas.
Necturus generally remain inactive throughout the summer. It is believed that they migrate to deeper waters where the water is cooler. There is speculation in the literature that Necturus may aestivate (Viosca 1937); however, this has not been proven either way. It is possible that Necturus simply seeks cooler waters and reduces activity during these periods as opposed to entering into a true aestivation. Necturus are most frequently observed in the colder months of the year, when water temperatures drop below 20°C (68°F). During this time, they may move as much as 1 km (0.6 miles) in a 24-hour period (Sajdak, 1982). They are nocturnal in nature, but may be more active during the day in deeper or muddy waters where the light levels are reduced.

N. m. maculosus |

N. m. maculosus |
Housing
One N. maculosus can be kept in a 20-gallon long (75x30x30 cm) or larger aquarium. Housing larger individuals or multiple adults together will require a larger space to prevent stress. They are territorial, and will fight if sufficient space and cover is unavailable. Territorial behaviors have been observed between two individuals housed in a 75-gallon (280-liter) tank, and subsequent fighting resulted in the ultimate separation of the individuals. Multiple cover objects should be provided. These may be pieces of driftwood, flowerpots, boards, rocks, or other objects, but make sure that the objects are free of chemicals, and lack sharp edges. Boards, driftwood, branches and/or logs from pine or cedar trees should be avoided due to the risk of poisoning from terpenes and other chemicals contained in the sap. Personal observations suggest that the animals prefer large flat rocks, especially when propped against another rock to create a cave-like area; these caves can be oriented toward the glass to make it easier to view the animal. Take care to ensure that the cover object cannot fall and injure the animal or break the glass of the aquarium should any rocks be disturbed. Mudpuppies themselves can move small rocks around easily. It is best to include two or three hiding spots per animal to allow each one to select a favorite spot.
Should an individual feel threatened, it will keep its gills close to its body. Defense of its own hiding spot generally consists of vigorous tail fanning across the entrance. Should another animal try to gain entrance, the resident mudpuppy may bite the intruder. If this kind of behavior is frequently observed in a multi-animal enclosure, separation or redesign of the aquarium is warranted.
Unlike many aquatic salamanders, N. maculosus can tolerate mild currents generated by filters. Current may even prove beneficial in creating a more natural stream-like environment. Mudpuppies can jump out of the enclosure, so a lid is required, unless the top of the enclosure is 10 cm or more above the water level. A screen lid is recommended, as the ventilation and evaporation can help reduce water temperature.
N. maculosus prefer temperatures below 20°C (68°F). In the winter, any temperature between 5 and 20°C (41-68°F) should be fine, though temperatures on the lower end of this range are strongly recommended. Summer temperatures should not exceed 22°C (72°F) for long periods of time. Anything over 25°C (77°F) may prove fatal. N. m. lousianensis may prove more tolerant of slightly higher temperatures than the northern race.
Stress on the animal should be kept to a minimum, as stressed animals are more likely to acquire fungal infection of the gills, which is usually fatal if untreated (personal observation). Many Necturus obtained from biological supply warehouses and similar commercial organizations perish from this sort of infection. Treatment usually involves the use of salt baths and/or medications and may require the assistance of a veterinarian. Gill size should also be monitored, as smaller gills may also signal an illness. No Necturus species undergoes metamorphosis, so gills will not shrink for this reason. Monitor gill size before attempting treatment, since size may fluctuate over time, or due to changes in water temperature and/or dissolved oxygen level.
 N. m. louisianensis |
 N. m. louisianensis |
Feeding
In the wild, N. maculosus generally eat crustaceans, insects, fish, annelids, mollusks, and amphibians (Petranka, 1998). In captivity, food items such as scuds (amphipods), aquatic insect larvae, snails, and worms are readily accepted. Crayfish are reported to be an important food item in the wild; however, in my own experience, live crayfish have been ignored. Fish to be used as feeders may be collected (check appropriate state regulations) or purchased from a pet store or supermarket. When purchasing fish for use as feeders, avoid using goldfish, as these are high in saturated fats. Preferably use livebearers, marine, or cold/cool water fish such as shiners, or rosy reds (a.k.a. fat-head minnows), as these are much lower in saturated fats and can contain beneficial polyunsaturated fats. To minimize the risk of disease transmission from fish, a few simple steps may be followed. The fish may be quarantined for 30 days and observed for parasites or external lesions, you may breed your own fish, and/or the fish can be frozen for two weeks or more to eliminate parasitic worms. However it should be noted that any collected food item(s) including earthworms or other annelids that are not frozen for at least two weeks prior to feeding could potentially transmit parasites. It should also be noted that freezing does not reduce the risk of the transmission of bacterial, viral, or fungal pathogens.
Quantity of food is dependent on frequency of feeding. For one adult, 3 large minnows or night crawlers 2-3 times a week is sufficient, adjusting for the size of the animal. A 12-inch (30 cm) adult can eat as many as 8 medium-sized minnows in a single feeding. The feeding schedule is very flexible; mudpuppies are adapted to long periods of time in the wild without food, and can maintain large fat reserves in order to survive. Adults may live for more than a month with very little food if the water temperature is cold. Personal observation has shown that feedings should be increased when the tail appears to be thinning.
Necturus will learn to take food from a keeper's hand, but care must be taken to avoid being bitten. To hand-feed, approach the animal slowly, and allow it to feed on its own terms. With patience, this can be one of the more rewarding aspects of keeping the species. Mudpuppies rarely bite, and a feeding accident is the most likely time for a bite to occur. Bites rarely break the skin, but can be painful. Teeth are designed for holding prey, not tearing, and food items are swallowed whole.
 N. m. louisianensis juvenile. |
 N. m. louisianensis subadult. |
Reproduction
N. maculosus mate in the fall/winter and lay eggs in the spring. Most populations deposit in late May to early June, though it may be as early as late April. Females construct nests on the underside of a cover object, and usually remain there until they hatch. It is unknown whether the female actively guards them, or merely continues to occupy the location. Regardless, the female's presence protects the eggs from predators such as fish or crayfish, and other mudpuppies. Females lay up to 12 eggs per day, in a single layer 15-30 cm (6-12 in) in diameter (Petranka 1998). Eggs are 5-6.5 mm (1/4 in) in diameter, and have three envelopes, with the outermost jelly coat allowing the eggs to adhere to the underside of the cover object. Clutch sizes vary, but are generally in the range of 60-100 eggs. Fertilization is internal, and sperm is retained for several months. Many other details about reproduction in this species are currently unknown.
Larvae hatch in July or August, after an estimated incubation period of 38-63 days. Larvae are much more colorful than adults, featuring a brown stripe lengthwise on a light body. They grow an average of 56 mm (2 in) per year in the wild, reaching maturity at 20 cm (8 in) total length, after approximately 5 years.
Gender in N. maculosus can be determined by examining the vent of the animal. During the breeding season, males will have a swollen cloaca, as well as a pair of nipple-like papillae at the rear of the vent (Sajdak, 1982). Additionally, the male vent has a crescent-shaped transverse groove. Females may have lighter coloration around the vent.
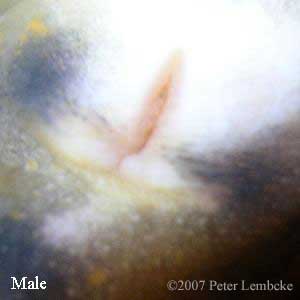 N. m. maculosus male vent. Note that the vent is more crescent shaped than the female, and the two papillae (small nubs) are visible at the lower end of the vent. |
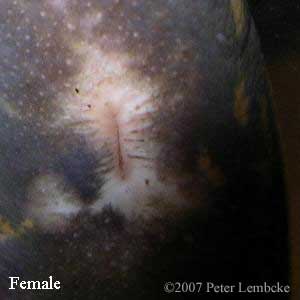 N. m. maculosus female vent. |
To date, there have been few reported captive breeding successes. The reasons for this may include their scarcity in captivity and the tank size requirements for keeping multiple animals together. A keeper wishing to attempt breeding should slowly begin decreasing the water temperature to around 15°C (59°F) or colder in the fall, and maintain this temperature until spring. It may be beneficial to manipulate the photoperiod in conjunction with temperature to maximize the chances of reproduction, but bright or intense lighting should be avoided as it may increase the stress on the animal. A strong current may be useful or even necessary; they are frequently observed downstream of dams in the wild trying to move upstream, so current orientation may play a role in the location of nesting sites and attraction of mates. Finally, introduction of unfamiliar animals at this time may also increase the chances of breeding.
 N. m. maculosus egg clutch. |
 Juvenile N. m. maculosus specimen from Michigan. |
Should breeding prove successful, it may be wise to remove all animals except for the nesting female from the enclosure, as adults have been known to cannibalize eggs. It is unknown if females will eat their own eggs. Despite this, leaving the eggs with the female may prove most successful; in almost all nesting caudate species studied to date, the removal of the female usually results in loss of many eggs to fungal infections. Artificial rearing of eggs is a labor-intensive method that requires the separation of the eggs into small groups, which are then monitored and agitated as if the female was attending the clutch. The highest artificial hatch rates have been obtained by the use of hatchers of the kind used by trout hatcheries (Vess, Harris; 1997). If a non-intrusive method of observation is possible, careful monitoring of the eggs and female may allow the keeper to salvage a nest if predation does occur.
One successful captive breeding has been reported (Raffaelli 2000). A group of 2:2 adults was housed in a tank 1.7 x 0.8 meters (5 x 2.6 feet) with a current. The temperature was cycled between 6°C in winter and 16°C in summer. A nuptial dance was observed in March, and eggs were found in May. Most of the eggs were lost to fungus, but best success was achieved for eggs left together with the female.
 Captive N. m. maculosus with eggs. |
 N. m. maculosus eggs laid in captivity. |
Conservation and Ecology
N. maculosus are abundant in many states. Specific populations may become threatened or extirpated due to siltation and/or pollution. They are considered threatened in Iowa, Indiana, Virginia, and Vermont, and vulnerable in Oklahoma. They are considered endangered in Georgia and Maryland. They may have been extirpated from South Dakota and North Carolina (NatureServe Explorer, 2004). Suitable habitat is decreasing in some areas, and other populations may begin declining in the future.
One major problem for Necturus is the lampricide, TFM. Areas experiencing problems with sea lampreys, which are a pest to the fishing industry, employ TFM to selectively kill lamprey larvae. Unfortunately, it also kills mudpuppies and other larval amphibians. In some areas treated with this chemical, populations have dropped drastically (Schmitt, 2004); however, the lampricide is unlikely to be solely responsible, as studies have shown that a dose exists that is lethal to lampreys but has limited effect on mudpuppies (Christie, 2000). All mudpuppies currently sold for pets or research are wild caught. They are used extensively for medical training and research. It is possible that commercial collecting may have an unsustainable impact on wild populations, especially in areas already under pressure from other threats.
N. maculosus is the only host of the salamander mussel. The salamander mussel uses the mudpuppy as a host during the mussel's larval stage. The mussel larvae attach themselves to the mudpuppy's gills, and drop off after metamorphosis (Michigan Natural Features Inventory, 2002). The relationship between the parasite and the host has not been extensively studied, but it is unlikely that the mussel kills the host animal. The salamander mussel is the only freshwater mussel that uses a non-fish host. This mussel is threatened or endangered in several states, and should Necturus become extirpated from an area, the mussel will likely follow, as the absence of mudpuppies will prevent successful reproduction of the mussel.
Predation of adult N. maculosus is rarely observed, though fish, turtles, and northern water snakes (Nerodia sipedon) have been documented as predators (Petranka, 1998). Adult mortality is presumably low and primarily attributed to human interference. Fishermen frequently catch them, and many times will leave them on the ice or on the shore, due to misconceptions about their predation of fish and fish eggs, the false belief that they are poisonous, or simply because of their general appearance. Little literature exists to document predation on young Necturus, but it can be assumed that fish, crayfish, wading birds, and other organisms eat them.
 Juvenile N. m. maculosus from Pennsylvania. |
 Juvenile N. m. maculosus from Pennsylvania. |
References
Christie, Gavin. 2000. Protecting Mudpuppies, Protecting Stream Ecosystems. Great Lakes Fishery Commission Forum: Fall 2000. Accessed 10 March 2005. http://www.glfc.org/pubs/Forum_F00.pdf
Michigan Natural Features Inventory. 2002 "Simpsonaias ambigua (Say): Salamander Mussel". Accessed 5 March 2005. http://web4.msue.msu.edu/mnfi/abstracts/aquatics/Simpsonaias_ambigua.pdf [link no longer available]
NatureServe Explorer. "Comprehensive Report: Necturus maculosus". Oct. 2004. Accessed 27 Feb 2005. http://explorer.natureserve.org/servlet/NatureServe?searchName=NECTURUS+MACULOSUS+
Petranka, James W. 1998. Salamanders of the United States and Canada. Smithsonian Institute Press: Washington.
Raffaelli, Jean. 2000. Observations on the reproduction of Necturus maculosus maculosus (Rafinesque) in captivity. Bull. Soc. Herp. Fr. 94: 25-27.
Sajdak, Richard A. 1982. "Seasonal activity patterns, habitat selection, and population structure of the Mudpuppy, Necturus maculosus, in a Wisconsin stream." MS Thesis; University of Wisconsin-Milwaukee.
Schmidt, Hunsinger, Coote, Griffin-Noyes, and Kiviat. 2004. The Mudpuppy (Necturus maculosus) in the Tidal Hudson River, with Comments on its Status as Native.Northeastern Naturalist. Accessed 10 March 2005. https://doi.org/10.1656/1092-6194(2004)011[0179:TMNMIT]2.0.CO;2
Vess, Tomalei J.; Harris, Reid N. 1997. Artificial brooding of salamander eggs, Herpetological Review 28(2): 80.
Viosca, P., Jr. 1937. A tentative revision of the genus Necturus with descriptions of three new species from the southern Gulf drainage area. Copeia 1937; 120-138.
©2005 Peter Lembcke
